Mary Madison, RN, RAC-CT, CDP
Clinical Consultant – Briggs Healthcare
- I blogged on the updated Interim Guidance for Rapid Antigen Testing for SARS-CoV-2 earlier this week. Thursday, August 20, 2020, AHCA/NCAL posted CDC Interim Guidance for Rapid Antigen Testing … AHCA/NCAL Member Summary (thank you so much!). “This document provides an explanation and summary of this guidance focusing on the elements that impact long-term care providers.”
This is an excellent 4-page summary that includes these categories:
- Definitions of Testing
- General Guidance
- Regulatory Requirements
- Specimen Collection
- Test Performance
- Evaluating the Results
- Reporting Results to Health Departments and Patients
- Additional Guidance from CDC
- Additional Testing Guidance from AHCA/NCAL
- Rapid Antigen Point of Care Devices- Training for Long Term Care Providers
The last 3 topics contain hyperlinks to additional information. Share this resource with your team and colleagues.
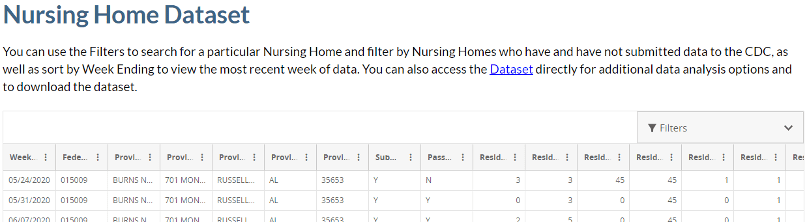
- COVID-19 Nursing Home Data for week ending August 9, 2020 is now available.

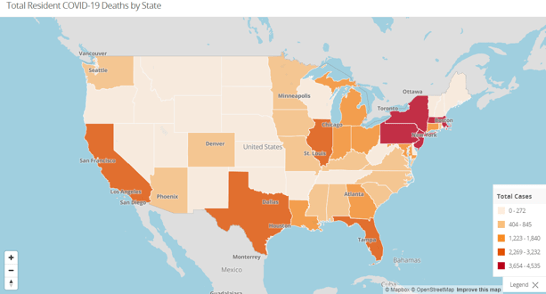
Scroll down this webpage to find:
- COVID-19 Emergency Declaration Blanket Waivers for Health Care Providers carries an update date of August 20, 2020. I’m not seeing any new information, but you’ll want to review and retain for your reference and records.
- The most recent Nursing Home Infection Control Surveys are now available.
