Mary Madison, RN, RAC-CT, CDP
Clinical Consultant – Briggs Healthcare
On November 30, 2020, the GAO (Government Accountability Office) issued a report to the U.S. Congress.
“This report examines the federal government’s continued efforts to respond to and recover from the COVID-19 pandemic and makes 11 new recommendations to federal agencies and raises one new matter for congressional consideration. Areas covered include medical supply shortages, COVID-19 testing, COVID-19 vaccines and therapeutics, nursing home care, assistance to individuals and businesses, and program integrity. This report includes 44 enclosures about a range of federal programs and activities across government, including the status of health care and economic indicators that could help monitor the nation’s response to and recovery from the COVID-19 pandemic, as well as its preparedness for future outbreaks (see app. I). Figure 1 lists these enclosures by topic area and highlights those with recommendations.”
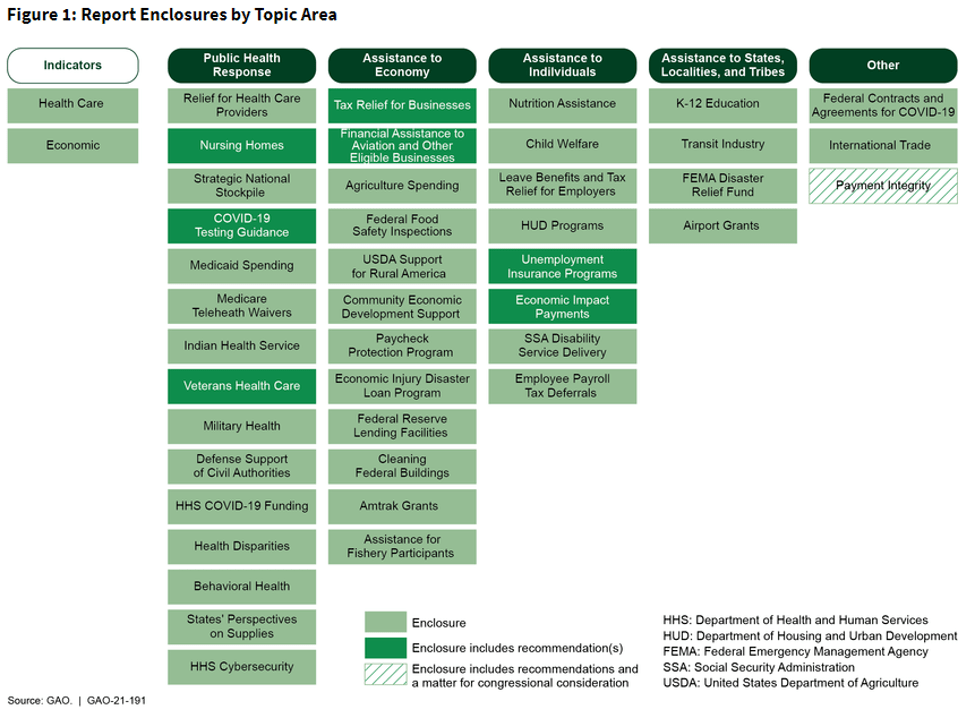
There’s a lot of real estate to navigate in this comprehensive report, to be sure! Grab a pot of coffee and settle in to digest all of the information provided in the report. You likely spotted that there are 44 enclosures alone in this report as well as 11 recommendations to federal agencies. (2nd paragraph of this blog). Appendix IV contains Comments from the Department of Health and Human Services. There’s also a 10.25-minute podcast to listen to. Share this report with your colleagues.
The report includes these statements regarding Nursing Homes (bolding added by me):
“To better inform its response, and that of other key stakeholders, to COVID-19 in nursing homes, we are recommending that CMS quickly develop a plan that further details how it intends to respond to and implement, as appropriate, the Nursing Home Commission’s recommendations. The plan should (1) include milestones that allow CMS to track and report on the status of each recommendation; (2) identify actions taken and planned, including areas where CMS determined not to take action; and (3) identify areas where CMS could coordinate with other federal and nonfederal entities. HHS neither agreed nor disagreed with our recommendation and said it would refer to and act upon the Commission’s recommendations, as appropriate.”
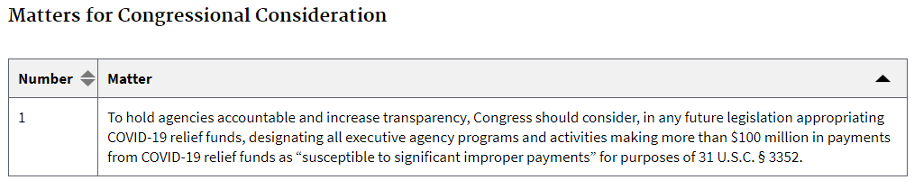
This recommendation is for Congress specifically: