Mary Madison, RN, RAC-CT, CDP
Clinical Consultant – Briggs Healthcare
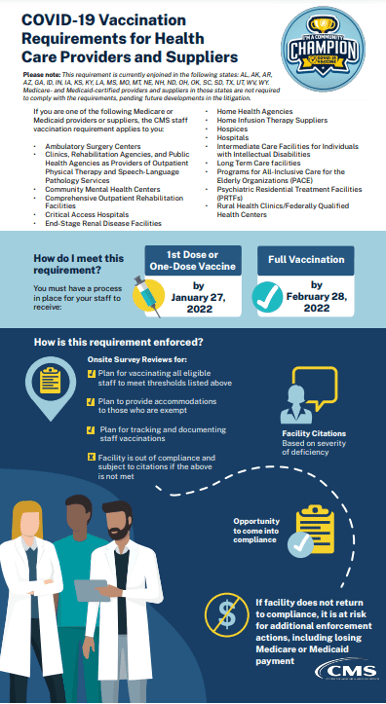
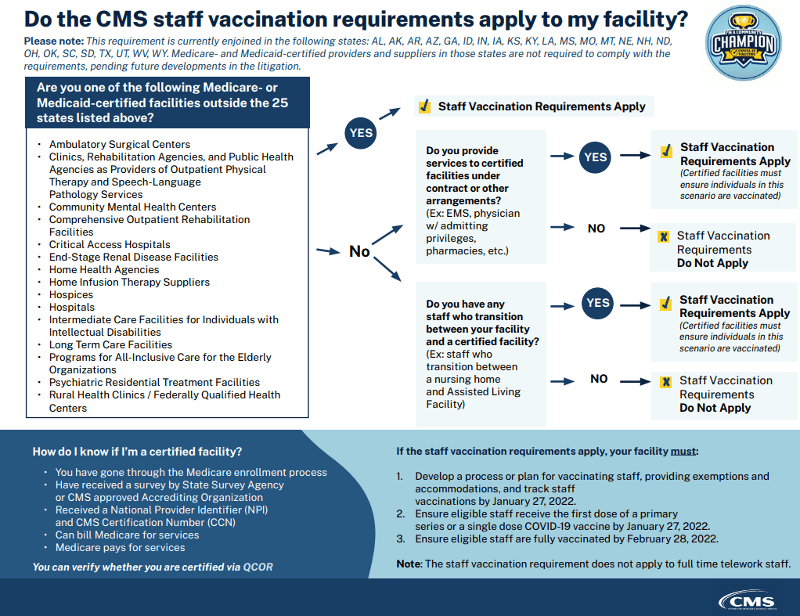
CMS posted a number of documents and infographics on Tuesday, December 28, 2021, related to staff vaccinations and the Interim Final Rule.
New as of 12/28/21
Q: With this guidance being issued today, when will enforcement of the IFC begin?
A: As of December 15, 2021, following decisions by the United States Court of Appeals for the Fifth, Eighth, and Eleventh Circuits and the United States District Court for the Northern District of Texas, implementation and enforcement of Medicare and Medicaid Programs; Omnibus COVID-19 Health Care Staff Vaccination, 86 Fed. Reg. 61,555, 61,556 (Nov. 5, 2021) (the “Interim Final Rule”), is preliminarily enjoined in the following twenty-five states: Alabama, Alaska, Arizona, Arkansas, Georgia, Idaho, Indiana, Iowa, Kansas, Kentucky, Louisiana, Mississippi, Missouri, Montana, Nebraska, New Hampshire, North Dakota, Ohio, Oklahoma, South Carolina, South Dakota, Texas, Utah, West Virginia, and Wyoming. Medicare- and Medicaid-certified providers and suppliers in those states are not required to comply with the Interim Final Rule, and surveyors will not investigate compliance with the rule in facilities located in those states, pending future developments in the litigation. In the other 25 states, the District of Columbia, and the territories, CMS as an exercise of enforcement discretion will begin monitoring and enforcing compliance on the following modified timeline: the deadline for Phase 1 is January 27, 2022, and the deadline for Phase 2 is February 28, 2022.
New as of 12/28/21 (I’ve provided a portion of the answer to this question below so be sure to read the entire answer in the FAQ.)
Q: Why is CMS enforcing the requirements of the IFC on a different timeline than the one described in the Interim Final Rule?
A: CMS therefore considers it in the best interests of public health to use its enforcement discretion to delay implementation of Phase 1 until January 27, 2022, for several reasons (described below).
First, CMS recognizes that it may be difficult to schedule vaccine administration appointments over the holidays, particularly in light of increased demand for booster doses and the possibility that facilities may also choose to provide post-administration sick leave to recipients. By delaying enforcement of Phase 1 until January 27, 2021, CMS expects that covered facilities will be better equipped to mitigate potential short-term workforce disruptions.
Second, CMS recognizes that facilities need time to restart and ramp up efforts to meet the requirements and that State Survey Agencies need time to restart and ramp up efforts to prepare to survey for compliance. The preliminary injunctions delayed issuance of guidance and deprived stakeholders of the opportunity to communicate with CMS. The agency has no desire to disadvantage facilities that might have been reliant on the availability of such guidance. CMS is nonetheless working to implement the IFC as expeditiously as reasonable in light of the ongoing urgency for the IFC.
CMS notes that its actions today merely describe its own timeline for enforcement and implementation and urges all facilities covered by the rule to come into compliance as soon as possible.